
Roskilde University
Jeppe Kari Lab
Research
Research topics
Jeppe Kari Lab aims to develop a kinetic theory for interfacial enzymes. We do this using 3 approaches:
Approach A
-
Adapting principles and practices from inorganic heterogeneous catalysis
Approach B
-
Adapting conventional enzyme kinetic theories and assays to heterogeneous and colloidal systems
Approach C
-
Development of in-silico-guided discovery and design of interfacial enzymes
A) Scaling relations
The application of empirical scaling relations such as Linear Free Energy Relationships (LFERs) is widely used in inorganic heterogeneous catalysis but essentially unexplored within heterogeneous biocatalysis. Jeppe Kari Lab seeks to explore the generality LFERs for interfacial enzymes. We have recently looked at this for enzymes that break down cellulose (cellulases). We aim to investigate whether other interfacial enzymes such as chitinases, amylases, and PETases may be governed by scaling relations.
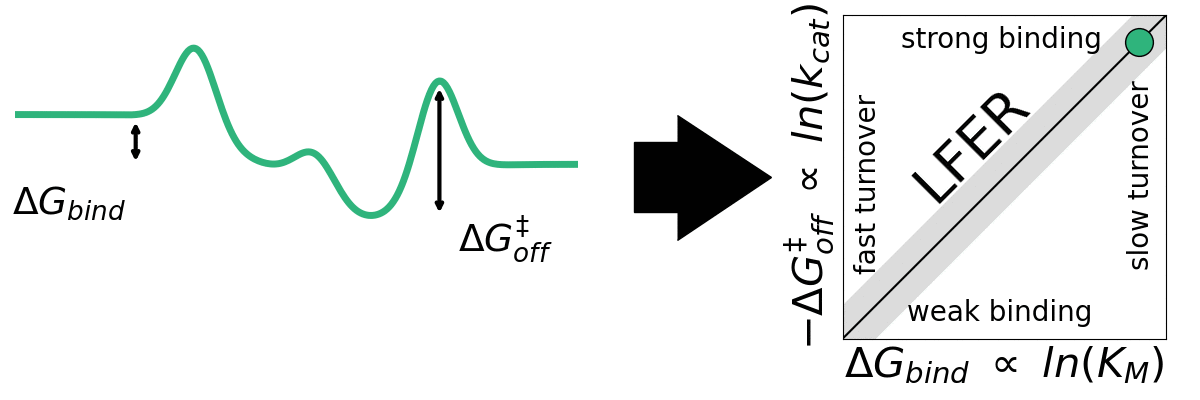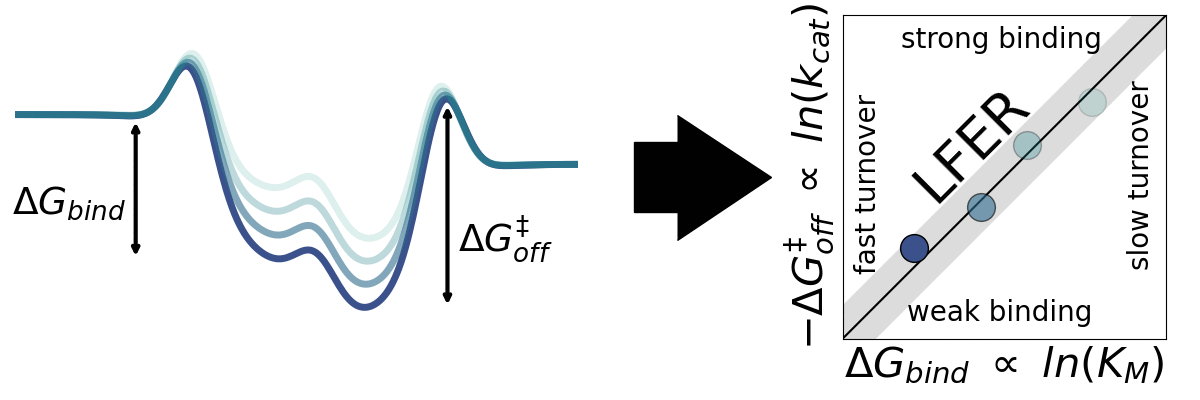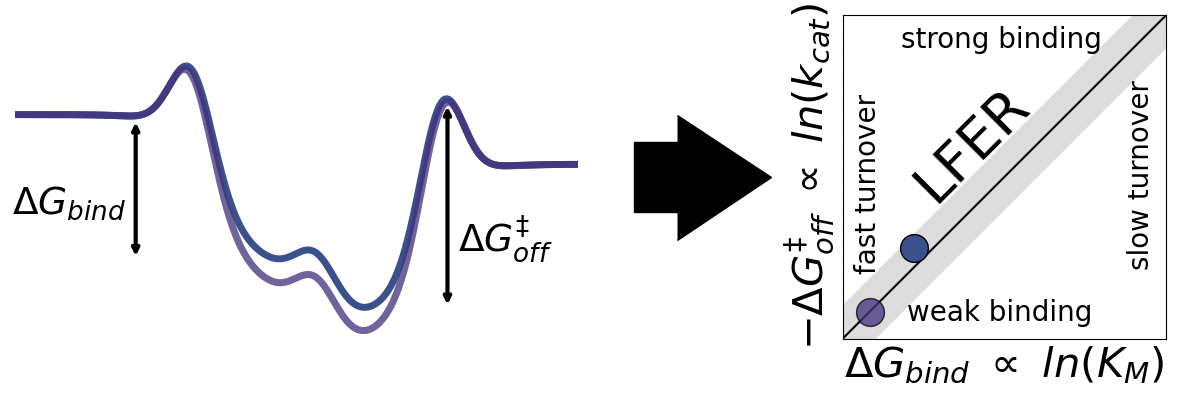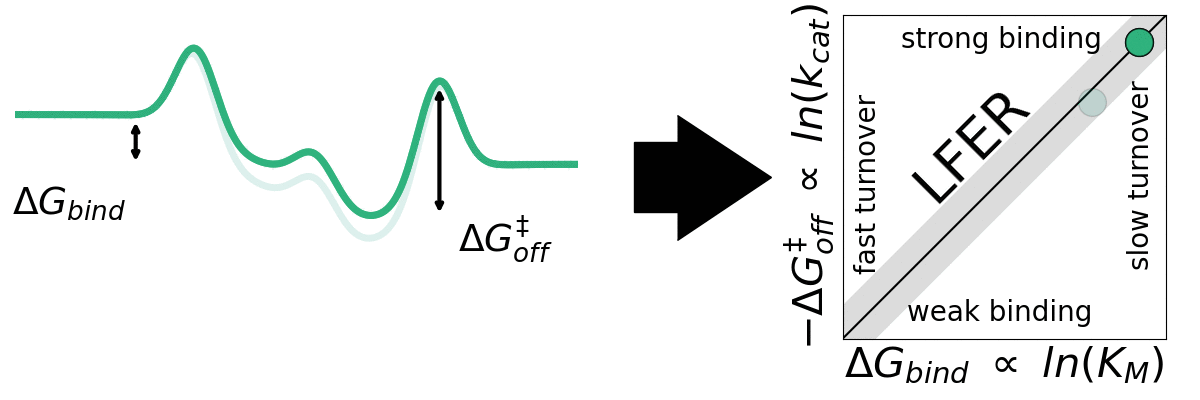
A) Sabatier principle
The Sabatier principle is a century old concept that remains one of the most fundamental tools in modern descriptions of heterogeneous catalysis. It states that efficient catalysis requires an intermediate interaction strength of the catalyst-reactant complex.
The Sabatier principle has proven tremendously useful as a tool to elucidate mechanisms, rate limiting steps, and as a guideline to design better technical catalysts. We have shown that the principle holds for interfacial enzymes such as PETase and cellulases and it can be used as a tool to guide enzyme design and discovery.

B) Interfacial enzyme kinetics
Kinetic information on interfacial enzymes acting on their real insoluble substrate are scarce. The solid substrate renders large-scale screening difficult so most kinetic studies on reel substrate are often done using a small set of enzymes under a limited set of experimental conditions. This lack of kinetic information limits advancement in the field as it hinders the development of reliable structure-activity relationships. For this reason we have developed assays and techniques to experimentally screen interfacial enzymes with (semi) high-throughput to expand the kinetic information of interfacial enzymes. A crucial part of this work is to find rational ways to screen these enzymes to extract descriptors with high functional information.

C) Virtual screening
A new design principle that combines computational methods (bioinformatics, homology modeling, MD simulation) with empirical trends. The central idea is to use scaling relations such as Linear Free Energy Relationships (LFERs) to reduce the engineering problem to a “1-dimensional” problem of affinity. This is attractive from a computational point of view since this may allow predictions of catalytic rates based solely on computed binding free energies. This approach may both be applied for in silico-driven discovery and as a design principle for engineering better industrial enzymes.

Heterogenous biocatalysis
Catalysis is traditionally divided into three categories; Homogeneous-, bio- and heterogeneous catalysis. Non-biochemical catalysis uses a fundamental distinction between homogenous and heterogeneous reactions. The former refers to cases where the catalyst and its reactant(s) are in the same phase (e.g. in solution), while the latter describes processes that occur at an interface, such as metals catalyzing the conversion of gaseous molecules. This distinction has proven tractable and is generally applied in all basic descriptions of conventional catalysis. An analogous distinction between bulk- and interfacial processes is rarely used in kinetic analyses of enzyme reactions in spite of the fact that interfacial reactions are widespread in both Nature and technical applications of enzymes.
The aim of Jeppe Kari Lab is to establish the field of heterogeneous biocatalysis (interfacial enzymology) by combining principles and practices from inorganic heterogeneous catalysis (approach A) and conventional (homogeneous) biocatalysis (approach B).

Selected Publications
Kari J. et al., ACS Catalysis 7 (7)
An Inverse Michaelis–Menten Approach for Interfacial Enzyme Kinetics
Kari J. et al., ACS Catalysis 8 (12)
Sabatier Principle for Interfacial (Heterogeneous) Enzyme Catalysis
Kari J. et al., Analytical Biochemistry 586
A practical approach to steady-state kinetic analysis of cellulases acting on their natural insoluble substrate
Kari J. et al., Biochemical Journal 477 (10)
A steady-state approach for inhibition of heterogeneous enzyme reactions
Kari J. et al., Nature Communication 12, 3847
Physical constraints and functional plasticity of cellulases
Bååth J. A. et al., JACS Au 2 (5)
Sabatier Principle for Rationalizing Enzymatic Hydrolysis of a Synthetic Polyester
Schaller K. S. et al., ACS Catalysis 12 (12)
Virtual Bioprospecting of Interfacial Enzymes: Relating Sequence and Kinetics